- Author Rachel Wainwright wainwright@abchealthonline.com.
- Public 2023-12-15 07:39.
- Last modified 2025-11-02 20:14.
Maxiflox
Maxiflox: instructions for use and reviews
- 1. Release form and composition
- 2. Pharmacological properties
- 3. Indications for use
- 4. Contraindications
- 5. Method of application and dosage
- 6. Side effects
- 7. Overdose
- 8. Special instructions
- 9. Application during pregnancy and lactation
- 10. Use in childhood
- 11. In case of impaired renal function
- 12. For violations of liver function
- 13. Use in the elderly
- 14. Drug interactions
- 15. Analogs
- 16. Terms and conditions of storage
- 17. Terms of dispensing from pharmacies
- 18. Reviews
- 19. Price in pharmacies
Latin name: Maxiflox
ATX code: S01AE07
Active ingredient: moxifloxacin (Moxifloxacin)
Manufacturer: K. O. Rompharm Company S. R. L. (SC Rompharm Company, SRL) (Romania)
Description and photo update: 2020-17-08
Prices in pharmacies: from 139 rubles.
Buy

Maxiflox is an antimicrobial drug from the group of fluoroquinolones for topical use in ophthalmology.
Release form and composition
Dosage form - eye drops, 5 mg / ml: greenish-yellow transparent solution (5 ml each in polyethylene bottles with a glued label, sealed with a dropper stopper with a lid equipped with a safety polyethylene ring; in a cardboard box 1 bottle and instructions for use Maxiflox).
Composition for 1 ml of the preparation:
- active substance: moxifloxacin - 5 mg (in the form of moxifloxacin hydrochloride - 5.45 mg);
- auxiliary components: boric acid - 3 mg; sodium chloride - 6.5 mg; 1 M hydrochloric acid solution / 1 M sodium hydroxide solution - up to pH 6.7-7.0; purified water - up to 1 ml.
Pharmacological properties
Pharmacodynamics
Maxiflox is an antimicrobial drug for topical use in ophthalmology with a wide spectrum of action. The mechanism of operation of its active component, moxifloxacin, fluoroquinolone with IV generation antibacterial activity, is due to the inhibition of topoisomerase IV and DNA gyrase, which recombine, replicate and repair deoxyribonucleic acid (DNA) in a bacterial cell.
The development of resistance to antibiotics of the fluoroquinolone series, including to moxifloxacin, occurs through chromosomal aberrations in the genes encoding topoisomerase IV and DNA gyrase. Moxifloxacin resistance in gram-negative bacteria is associated with mutations in multiple antibiotic and quinolone resistance systems. The development of antibiotic resistance is also associated with the expression of efflux pumps (proteins in the cell membrane) and inactivating bacterial enzymes. Cross-resistance with aminoglycosides, macrolides and tetracyclines is not expected due to differences in their mechanism of action. The development of resistance can be influenced by factors such as geographic location and time of year, therefore it is important to obtain information on antibiotic resistance in a specific area before starting therapy,which is especially important in the treatment of severe infections.
Maxiflox is active against most strains of microorganisms (both in vitro and in vivo):
- gram-positive bacteria: Corynebacterium spp. (including Corynebacterium diphtheriae), Micrococcus luteus (including strains insensitive to gentamicin, tetracycline, trimethoprim and erythromycin), Staphylococcus aureus (including those insensitive to methicillin, gentamycin, trimethoprimicin, ofloxacin erythromycin strains), Staphylococcus epidermidis (including those insensitive to methicillin, gentamicin, ofloxacin, tetracycline, trimethoprim, erythromycin strains), Staphylococcus haemolyticus (including those insensitive to methicillin, ofmethocycin, gentamycinam), Staphylococcus hominis (including strains insensitive to methicillin, erythromycin, tetracycline, trimethoprim), Staphylococcus warneri (including strains insensitive to erythromycin), Streptococcus mitis (incl.strains insensitive to penicillin, tetracycline, trimethoprim, erythromycin), Streptococcus pneumoniae (including those insensitive to penicillin, gentamycin, tetracycline, trimethoprim, erythromycin strains), Streptococcus tricycin, pentacillus viridans (incl. erythromycin strains);
- gram-negative bacteria: Acinetobacter lwoffii, Haemophilus influenzae (including ampicillin-insensitive strains), Haemophilus parainfluenzae, Klebsiella spp.;
- other microorganisms: Chlamydia trachomatis.
Moxifloxacin acts in vitro against most of the microorganisms listed below, but the clinical significance of these findings is unknown:
- gram-positive bacteria: Listeria monocytogenes, Staphylococcus saprophyticus, Streptococcus agalactiae, Streptococcus mitis, Streptococcus pyogenes, Streptococcus groups C, G, F;
- gram-negative bacteria: Acinetobacter calcoaceticus, Acinetobacter baumannii, Enterobacter cloacae, Escherichia coli, Enterobacter aerogenes, Citrobacter freundii, Citrobacter koseri, Klebsiella pneumoniae, Klebsiella oxytoca, Morganella morganser, Protezerisella vulorhoe, Proteinisella gum, Proteiniselle gum
- anaerobic microorganisms: Fusobacterium spp., Clostridium perfringens, Prevotella spp., Propionibacterium acnes, Prevotella spp.;
- other microorganisms: Chlamydia pneumoniae, Mycobacterium avium, Legionella pneumophila, Mycoplasma pneumoniae, Mycobacterium marinum.
There are no data on the relationship between the clinical and bacteriological outcome of moxifloxacin therapy for infectious diseases of the organ of vision.
Epidemiological data published by the European Antimicrobial Susceptibility Committee - Moxifloxacin inhibitory concentrations (cut-off values) for various microorganisms:
- Staphylococcus aureus - 0.25 mg / l;
- Coagulase-negative staphylococci - 0.25 mg / l;
- Streptococcus pyogenes - 0.5 mg / l;
- Streptococcus pneumoniae - 0.5 mg / l;
- Streptococcus, viridans group - 0.5 mg / l;
- Haemophilus influenzae - 0.125 mg / l;
- Enterobacter spp. - 0.25 mg / l;
- Klebsiella spp. - 0.25 mg / l;
- Morganella morganii - 0.25 mg / l;
- Moraxella catarrhalis - 0.25 mg / l;
- Neisseria gonorrhoeae - 0.032 mg / l;
- Serratia marcescens - 1 mg / l;
- Pseudomonas aeruginosa - 4 mg / l;
- Corynebacterium - no data available.
Pharmacokinetics
When applied topically, after instillation of Maxiflox eye drops, systemic absorption of moxifloxacin occurs.
In clinical studies, patients, 21 participants of both sexes, received moxifloxacin in the form of eye drops in both eyes three times a day, 1 drop for 4 days. The mean maximum plasma concentration (C max) of moxifloxacin at steady state was 2.7 ng / ml, the area under the concentration-time curve (AUC) was 41.9 ng / h / ml. The results obtained are approximately 1600 times (for C max) and 1200 times (for AUC) lower than the average values of these indicators after oral administration of moxifloxacin at a therapeutic dose of 400 mg.
T 1/2 (half-life) of moxifloxacin is ~ 13 hours.
Indications for use
Maxiflox eye drops are used to treat bacterial conjunctivitis caused by microorganisms sensitive to moxifloxacin.
Contraindications
It is contraindicated to prescribe Maxiflox to patients with established individual hypersensitivity to drugs of the quinolone series, any components in the composition of eye drops, as well as infants under 1 year of age.
With caution, only under the supervision of a specialist, provided that the expected therapeutic effect is exceeded over the potential risk to the fetus and child, it is recommended to use the drug during pregnancy and during breastfeeding.
Maxiflox, instructions for use: method and dosage
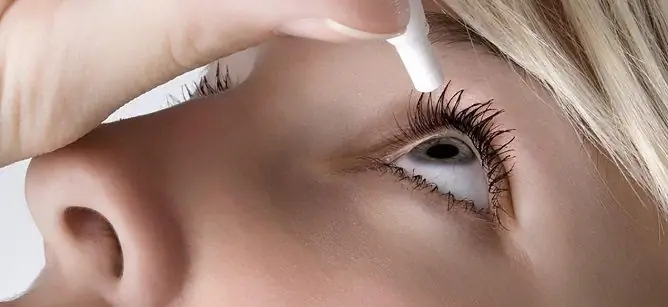
Maxiflox eye drops are applied topically, by instillation into the conjunctival sac of the affected eye.
It is necessary to carry out therapy taking into account the official recommendations for the use of antibacterial drugs. Maxiflox is intended for local ophthalmic use; the drug cannot be used as subconjunctival injections or for administration into the anterior chamber of the eye.
Adult patients (including elderly people over 65 years old) are recommended to instill 1 drop into the affected eye three times a day. Improvement of the condition is observed after 5 days from the beginning of the course, but therapy should be continued for another 2-3 days.
In the absence of a therapeutic effect from therapy after 5 days, it is required to verify the diagnosis and revise the treatment regimen. The duration of the therapeutic course depends on the severity of the disease, the clinical and bacteriological specificity of the course of the infectious process.
Correction of the dosage regimen of Maxiflox is not required when using the drug in pediatric practice.
Patients with hepatic and renal impairment do not require dose adjustment of moxifloxacin.
To prevent microbial contamination of the tip of the dropper bottle and the solution contained in it, during instillation, avoid contact with the eyelids, eyelashes and skin of the periorbital zone, as well as other surfaces.
To prevent the absorption of the solution after instillation through the mucous membrane of the nasal cavity, it is necessary to pinch the nasolacrimal canal with a finger for 2-3 minutes.
In the case of prescribing several ophthalmic preparations for topical use, the interval between their use should be at least 5 minutes, while the eye ointments are placed behind the eyelid into the conjunctival sac last.
Side effects
General information about the safety profile of moxifloxacin in the dosage form of eye drops was obtained in clinical studies in ophthalmology in 2252 patients who received therapy at a dose of 1 drop up to eight times a day. At the same time, 1900 people from the study group received moxifloxacin in the 1 drop regimen three times a day. The safety assessment was carried out in a mixed population, in which 1389 patients from the USA and Canada participated, 586 from Japan, and 277 from India. generally not received. The most commonly reported treatment-related adverse reactions were eye irritation and eye pain. The total frequency of their occurrence varied within 1-2% and in 96 cases out of 100 it was attributed to mild severity. At the same time, in one of the volunteers who took part in the study, the severity of the undesirable action led to the exit from the research program.
To classify undesirable side reactions, the following gradation of frequency of occurrence was used: very often - more than 1/10; often - more than 1/100, but less than 1/10; infrequently - more than 1/1000, but less than 1/100; rarely - more than 1/10 000, but less than 1/1000; very rarely - less than 1/10 000; the frequency is unknown - it is impossible from the available data to establish the frequency of the development of side effects.
System-organ classification of Maxiflox adverse reactions in accordance with the above gradation of frequency of occurrence:
- blood and lymphatic system: rarely - a decrease in the concentration of hemoglobin;
- immune system: frequency unknown - hypersensitivity;
- nervous system: infrequently - headache; rarely - paresthesia; frequency unknown - dizziness;
- organ of vision: often - eye pain, eye irritation; infrequently - dry eye syndrome, punctate keratitis, itchy eyes, subconjunctival hemorrhage, conjunctival injection, eye discomfort, eyelid edema; rarely - epithelial corneal defects, corneal injuries, blepharitis, conjunctivitis, conjunctival edema, decreased visual acuity, blurred vision, eyelid erythema, asthenopia; frequency unknown - ulcerative keratitis, endophthalmitis, corneal erosion, increased intraocular pressure, corneal opacity / edema / infiltrates, corneal tissue deposits, eye allergic responses, photophobia, lacrimation, keratitis, discharge from the eyes, foreign body sensation in the eye;
- heart: frequency unknown - palpitations;
- respiratory system, organs of the chest and mediastinum: rarely - pain in the pharynx and larynx, discomfort in the nose, feeling of the presence of a foreign body in the throat; frequency unknown - shortness of breath;
- digestive tract: infrequently - dysgeusia; rarely - vomiting; frequency unknown - nausea;
- hepatobiliary system: rarely - increased levels of AMT (aminotransferase) and GGT (gamma glutamyltransferase);
- skin and subcutaneous fat: frequency unknown - rash, urticaria, erythema, pruritus.
In patients receiving systemic therapy with fluoroquinolones, in rare cases, ruptures of the tendons of the joints of the arms, shoulder joint, Achilles tendon, as well as breaks of other tendons were observed. Such injury led to long-term disability or required surgery. According to the results of clinical studies and observations in the post-registration period, it was found that when receiving systemic treatment with fluoroquinolones, the risk of such injuries may increase if corticosteroids are included in the therapeutic regimen, and elderly patients are at particular risk. Mostly, there were ruptures of the tendons of the supporting joints, including the Achilles tendon.
Clinical studies of the use of moxifloxacin in the form of instillations were carried out with the participation of children, including newborns, as a result of which the similarity of the safety profile in the pediatric population with that in adult patients was confirmed. In children and adolescents under 18 years of age, eye irritation and eye pain were most often observed (frequency of occurrence ~ 0.9%). There were no differences in the profile of unwanted actions and their severity in children compared with the adult audience.
Overdose
Due to the small capacity of the conjunctival cavity, the likelihood of a local overdose of moxifloxacin when used in the form of eye instillations is virtually absent.
The total amount of moxifloxacin in the solution and the volume of the vial are too small for the development of undesirable side effects in the event of unintentional swallowing of the contents of the vial.
special instructions
With the systemic use of drugs of the quinolone series, severe allergic reactions, such as anaphylaxis, may develop, in some cases with a fatal outcome. Sometimes severe life-threatening hypersensitivity reactions to moxifloxacin occur even after the first dose. Some of them are accompanied by loss of consciousness, collapse, Quincke's edema (including swelling of the pharynx, larynx or face), shortness of breath, airway obstruction, urticaria, and itchy skin. If any symptoms appear that confirm the patient's increased sensitivity to Maxiflox, therapy should be discontinued. Severe hypersensitivity reactions to moxifloxacin and / or other components of the solution in an acute course may require urgent resuscitation, including oxygen therapy with airway control,according to indications.
Long-term use of eye drops can provoke excessive growth of microflora resistant to moxifloxacin, including excessive growth of fungi. With the development of superinfection, Maxiflox must be canceled and appropriate treatment prescribed.
Inflammation and rupture of tendons were observed with systemic use of drugs from the group of fluoroquinolones, primarily in elderly patients, as well as with their combined use with corticosteroids. In ophthalmology, after topical application, the systemic concentrations of moxifloxacin are significantly lower than those when the drug is taken orally, but when the first symptoms of tendinitis are diagnosed, instillation of eye drops should be stopped immediately.
There is insufficient information on the efficacy and safety of topical application of moxifloxacin in the treatment of bacterial conjunctivitis in newborns. As a result, it is not recommended to use Maxiflox for this disease in patients of this age category.
Due to the presence of a large number of Neisseria gonorrhoeae strains resistant to moxifloxacin, Maxiflox is not used for ex juvantibus therapy (empirical treatment) or prevention of gonococcal conjunctivitis, including the treatment of neonatal ophthalmia of gonococcal etiology. For Neisseria gonorrhoeae eye infections, patients should receive appropriate systemic treatment.
Due to the fact that there is insufficient information on the efficacy and safety of topical application of moxifloxacin in children under 2 years of age, it is not recommended to use Maxiflox in this category of patients for the treatment of eye infections caused by Chlamydia trachomatis. The use of the drug for the treatment of patients over 2 years of age with eye diseases caused by Chlamydia trachomatis should be combined with systemic therapy.
Treatment of neonatal conjunctivitis is carried out after a series of serological and bacteriological studies to determine the causative agent of the infection. According to the results of the examination, the treatment corresponding to the child's condition is established. For ophthalmia of newborns of chlamydial and gonorrheal etiology, systemic therapy is prescribed.
Contact lenses are not recommended for patients with infectious diseases of the anterior segment of the eyeball.
Influence on the ability to drive vehicles and complex mechanisms
As with the instillation of other eye drops, after the application of Maxiflox, a temporary loss of visual clarity is possible. Therefore, patients should refrain from driving vehicles or working with machines, complex machinery and production equipment until vision is fully restored.
Application during pregnancy and lactation
There is insufficient data on the use of moxifloxacin during pregnancy and during breastfeeding. In this regard, Maxiflox eye drops should be used for the treatment of pregnant and breastfeeding women with caution when the expected therapeutic effect outweighs the potential risks to the fetus / child.
In experiments carried out on animals, it was found that after taking moxifloxacin, a small amount of the substance passes into breast milk. In the case of careful adherence to the dosage regimen during the instillation of therapeutic doses of Maxiflox, the development of negative adverse reactions in infants is not expected.
In the course of preclinical studies of the teratogenic effect of moxifloxacin in animals, it was found that it did not adversely affect the embryonic development of the fetus when taken orally at doses of 500 mg / kg / day (which is 21,700 times more than the recommended daily dose for humans). But at the same time, there was a slight decrease in fetal weight and a delay in the development of the musculoskeletal system. While taking moxifloxacin orally at a dose of 100 mg / kg / day, the incidence of decreased growth in newborns increased.
Studies on the effect of Maxiflox instillations on human fertility have not been conducted.
Pediatric use
In pediatric practice, the use of Maxiflox is contraindicated for the treatment of infants under the age of 12 months.
When treating children over 1 year of age, no dosage adjustment is required.
With impaired renal function
No dose adjustment of Maxiflox is required for patients with impaired renal function.
For violations of liver function
No dose adjustment of Maxiflox is required for patients with impaired hepatic function.
Use in the elderly
The method of administration and dose of Maxiflox in elderly patients over 65 years old are similar to those in younger patients.
Elderly patients receiving systemic therapy with fluoroquinolones are at a particular risk of tendon ruptures (primarily the tendons of the supporting joints and the Achilles tendon), which can increase with the inclusion of corticosteroids in the treatment regimen.
Drug interactions
Since the low systemic concentration as a result of topical application of moxifloxacin in the form of instillations makes the pharmacological interaction of Maxiflox eye drops with other drugs / agents unlikely, its studies have not been conducted.
Analogs
Analogs of Maxiflox are Bivox VM, Vigamox, Dancil, Zimar, Levofloxacin-Optic, Lofox, Moxifur, Ofloxacin, Oftaquix, Signicef, Uniflox, Floxal, Ciprolet, Ciprolon, Cipromed, Ciprofloxacin and others, Ciprofloxacin.
Terms and conditions of storage
Store in original packaging at temperatures up to 25 ° C. Keep out of the reach of children.
The shelf life of drops is 3 years.
After opening, the contents of the bottle can be used for no more than 4 weeks. After the expiration date, the solution cannot be used.
Terms of dispensing from pharmacies
Dispensed by prescription.
Reviews about Maxiflox
According to reviews, Maxiflox is a very effective, high quality and inexpensive antibiotic for treating eye infections. Patients also note a convenient application regimen and a short treatment course.
Some complain about the lack of drops in pharmacies. In addition, in some cases, they describe adverse reactions in the form of pain and irritation in the eyes.
Price for Maxiflox in pharmacies
The approximate price of Maxiflox, eye drops, 5 mg / ml, for a dropper bottle containing 5 ml of solution, can range from 139 to 173 rubles.
Maxiflox: prices in online pharmacies
|
Drug name Price Pharmacy |
|
Maxiflox 5 mg / ml eye drops 5 ml 1 pc. 139 RUB Buy |

Anna Kozlova Medical journalist About the author
Education: Rostov State Medical University, specialty "General Medicine".
Information about the drug is generalized, provided for informational purposes only and does not replace the official instructions. Self-medication is hazardous to health!