- Author Rachel Wainwright wainwright@abchealthonline.com.
- Public 2023-12-15 07:39.
- Last modified 2025-11-02 20:14.
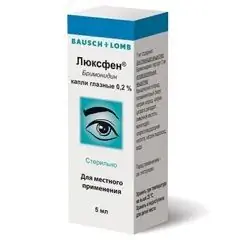
Luxfen
Luxfen: instructions for use and reviews
- 1. Release form and composition
- 2. Pharmacological properties
- 3. Indications for use
- 4. Contraindications
- 5. Method of application and dosage
- 6. Side effects
- 7. Overdose
- 8. Special instructions
- 9. Application during pregnancy and lactation
- 10. Use in childhood
- 11. In case of impaired renal function
- 12. For violations of liver function
- 13. Use in the elderly
- 14. Drug interactions
- 15. Analogs
- 16. Terms and conditions of storage
- 17. Terms of dispensing from pharmacies
- 18. Reviews
- 19. Price in pharmacies
Latin name: Luxfen
ATX code: S01EA05
Active ingredient: brimonidine (Brimonidine)
Producer: Santonika, JSC (SANTONIKA) (Lithuania)
Description and photo updated: 2018-29-11
Prices in pharmacies: from 309 rubles.
Buy
Luxfen is an anti-glaucoma agent.
Release form and composition
Dosage form - eye drops: transparent solution of greenish-yellow color (5 ml in polyethylene bottles with first opening control, equipped with a dropper nozzle, in a cardboard box 1 bottle and instructions for use of Luxfen).
Composition of 1 ml drops:
- active substance: brimonidine tartrate - 2 mg;
- auxiliary components: purified water, sodium citrate dihydrate, sodium chloride, polyvinyl alcohol, citric acid monohydrate, benzalkonium chloride, 1M sodium hydroxide solution or 1M hydrochloric acid solution.
Pharmacological properties
Pharmacodynamics
The active substance of Luxfen, brimonidine, is a selective antagonist of α 2 -adrenergic receptors. A decrease in intraocular pressure (IOP) occurs due to a dual mechanism of action: due to a decrease in the synthesis of intraocular fluid and an increase in uveoscleral outflow.
After instillation of eye drops, the decrease in IOP is 10-12 mm Hg. Art. The maximum effect of the drug develops within 2 hours and lasts for 12 hours.
Pharmacokinetics
The maximum plasma concentration (C max) of brimonidine after instillation of Luxfen is achieved in 0.5-2.5 hours. After applying eye drops 2 times a day for 10 days, the average C max remains low (approximately 0.06 ng / ml). When applied topically, about 29% of the substance binds to plasma proteins. Systemic absorption after instillation of the drug into the eyes is slowed down.
Brimonidine binds reversibly to melanin. Its concentration in the iris, retina and ciliary body after 2 weeks of using Luxfen is 3-17 times higher than the same indicators after a single instillation of eye drops.
Brimonidine is metabolized mainly in the liver. The substance itself and its metabolites are excreted by the kidneys. The half-life is about 2 hours.
Indications for use
- open-angle glaucoma;
- ophthalmic hypertension (as a monopreparation or in combination with other drugs that reduce intraocular pressure).
Contraindications
Absolute:
- children under 2 years old;
- lactation period;
- concomitant use of tricyclic antidepressants or monoamine oxidase (MAO) inhibitors;
- hypersensitivity to any component of the drug.
Relative (Luxfen eye drops should be used with caution):
- insufficiency of cerebral circulation;
- depression;
- orthostatic hypotension;
- heart failure;
- thromboangiitis obliterans;
- Raynaud's syndrome;
- renal failure (creatinine clearance <40 ml / min);
- liver failure;
- children and adolescents from 2 to 18 years old;
- the period of pregnancy (only when the benefits of therapy for the mother outweigh the possible risks to the fetus);
- simultaneous use of cardiac glycosides or antihypertensive drugs.
Luxfen, instructions for use: method and dosage
Luxfen eye drops should be instilled into the conjunctival sac of the affected eye.
Recommended dosing regimen: 1 drop 2 times a day (at 12-hour intervals). The course of therapy is determined by the doctor individually.
To minimize systemic absorption of the drug, it is recommended to squeeze the lacrimal sac at the inner corner of the eye immediately after instillation for 1 minute (closing the lacrimal opening).
Side effects
The most common side effects from the organ of vision are itching of the mucous membrane of the eyes, conjunctival hyperemia, allergic conjunctivitis, itching of the eyelid skin. Most of them, as a rule, are expressed in a mild degree and have a transient character, therefore, do not require the abolition of Luxfen.
According to the data of clinical studies, allergic reactions from the eyes were observed in 12.7% of cases (discontinuation of therapy was required in 11.5% of cases), occurred mainly after 3-6 months of treatment.
Possible side effects (classified by frequency of development as follows: very often - ≥ 1/10, often - from ≥ 1/100 to <1/10, infrequently - from ≥ 1/1000 to <1/100, rarely - from ≥ 1 / 10,000 to <1/1000, very rarely - <1/10 000):
- on the part of the organ of vision: very often - a burning sensation or a foreign body in the eye, conjunctival hyperemia, blurred vision, itching of the eyelids and mucous membranes of the eyes, follicular conjunctivitis, allergic conjunctivitis, allergic blepharitis, allergic blepharoconjunctivitis; often - mucous discharge from the eyes, conjunctival edema, hyperemia and edema of the eyelids, conjunctivitis, blepharitis, pain in the eye and lacrimation, retention lacrimation, folliculosis of the conjunctiva, eyelid lesions, photophobia, decreased visual acuity, blanching of the conjunctiva, mucous membrane, dryness and dryness, loss of visual field, photosensitivity, epiphora, floating opacities in the vitreous body, superficial spotty keratopathy, local allergic reactions of the mucous membrane of the eyes (including keratoconjunctivitis),functional damage to the vitreous body, hemorrhage in the conjunctiva or vitreous body, corneal opacity, erosion, keratitis, cataract; infrequently - barley; very rarely - miosis, iritis;
- from the immune system: infrequently - systemic allergic reactions;
- from the nervous system: very often - drowsiness, headache; often - a violation of taste, dizziness; infrequently - depression; very rarely - insomnia, fainting;
- from the digestive system: very often - dryness of the oral mucosa; often - dyspeptic disorders;
- from the respiratory system: often - pharyngitis, shortness of breath, cough, bronchitis; infrequently - dryness of the nasal mucosa;
- on the part of the cardiovascular system: infrequently - a feeling of palpitations, arrhythmias (including bradycardia and tachycardia); very rarely - an increase or decrease in blood pressure;
- on the part of the skin: often - rash;
- on the part of laboratory indicators: often - hypercholesterolemia;
- infectious and parasitic diseases: often - rhinitis, flu-like syndrome, sinusitis (including infectious), infectious disease (chills and respiratory infection);
- others: very often - fatigue; often asthenia.
In children, the following adverse reactions are possible: drowsiness, pallor, decreased attention, hypothermia, apnea, muscle hypotension, decreased blood pressure, bradycardia.
In the course of post-marketing studies, other side effects have been established, however, the reports were spontaneous, so it is not possible to establish the frequency of occurrence of reactions. These include:
- on the part of the organ of vision - iridocyclitis (anterior uveitis);
- from the digestive system - nausea;
- on the part of the skin: allergic reactions, including rash, itching, facial edema, erythema, vasodilation of the eyelid skin vessels.
Overdose
In case of an overdose with topical application, it is possible to develop / intensify the reactions described in the side effects.
In case of accidental ingestion of eye drops inside, adults may experience the following symptoms: drowsiness, depression of the central nervous system, depression or loss of consciousness, asthenia, cyanosis of the skin, a decrease in body temperature, vomiting, bradycardia, arrhythmia, hypotension, miosis, convulsions, apnea. Symptomatic and supportive therapy is performed. If necessary, provide airway patency.
In children, when using Luxfen as an adjunctive therapy for congenital glaucoma, as well as in case of accidental ingestion of eye drops inside, the following symptoms are possible: drowsiness, lethargy, pallor, loss of consciousness, hypothermia, cyanosis, hypotension, hypotension, bradycardia, respiratory depression and apnea. Conduct symptomatic and supportive therapy, provide airway control. In some cases, intensive care with intubation is required.
special instructions
The benzalkonium chloride contained in the preparation as a preservative can irritate the eyes.
Patients who use contact lenses should avoid getting solution on them, as this is fraught with discoloration. In this regard, before instilling Luxfen, the lenses should be removed, they are allowed to be reinstalled no earlier than 15 minutes later.
If allergic reactions develop during therapy, you should stop using Luxfen and consult a doctor. With the development of a delayed-type hypersensitivity reaction, intraocular pressure may increase.
Luxfen may increase the severity of the course of diseases caused by vascular insufficiency, such as depression, cerebrovascular accident, thromboangiitis obliterans, Raynaud's syndrome, orthostatic hypotension, heart failure.
If necessary, the simultaneous use of other ophthalmic agents between their instillations should be observed at intervals of 5-15 minutes.
Careful observation during the treatment period is required for children 2-7 years old (especially those with a body weight of less than 20 kg), since they often develop such a side effect as drowsiness, due to which in some cases it is necessary to cancel Luxfen. The incidence of drowsiness may decrease with age, but depends mainly on body weight.
Influence on the ability to drive vehicles and complex mechanisms
Luxfen often causes a disturbance in the clarity of visual perception, a state of drowsiness and fatigue. In this regard, patients are advised to refrain from driving vehicles and work with potentially dangerous consequences during treatment.
Application during pregnancy and lactation
Animal studies have not shown any negative effect of the drug on the intrauterine development of the fetus. However, the safety of brimonidine when used topically during pregnancy in humans has not been determined. For this reason, Luxfen is prescribed to pregnant women only in justified cases, when the benefits of therapy for the mother outweigh the possible risks to the fetus.
It is not known whether brimonidine, used conjunctivally, penetrates into breast milk. However, the likelihood cannot be completely ruled out, therefore Luxfen is contraindicated during lactation.
Pediatric use
- up to 2 years: the use of the drug is contraindicated;
- 2-18 years: Luxfen should be used with caution.
With impaired renal function
In renal failure (creatinine clearance <40 ml / min), Luxfen should be used with caution for treatment.
For violations of liver function
With hepatic insufficiency, use Luxfen for treatment with caution.
Use in the elderly
Elderly patients do not need to adjust the dose.
Drug interactions
Studies of possible interactions of Luxfen with other drugs have not been conducted.
Brimonidine is contraindicated in patients receiving MAO inhibitors, tri- or tetracyclic antidepressants (including mianserin).
A solution of brimonidine at a concentration of 2 mg / ml can enhance the effects of substances that affect the central nervous system, such as sedatives, opioids, barbiturates, anesthetics, ethanol.
In some patients, while using Luxfen, a slight decrease in blood pressure occurs, therefore, caution should be exercised when prescribing antihypertensive drugs and / or cardiac glycosides in combination.
Careful observation is necessary for patients who are simultaneously taking drugs that can affect the absorption and metabolism of adrenaline, norepinephrine and other so-called biogenic amines (reserpine, methylphenidate, chlorpromazine).
With the first systemic use of agonists or antagonists of adrenergic receptors (for example, isoprenaline or prazosin) and a change in the dose of these drugs, regardless of the method of their administration, it is possible to develop drug interactions with α-adrenergic receptor agonists (including brimonidine) and, as a consequence, change their effects.
Analogs
Luxfen's analogs are: Alfabrim, Azarga, Alfagan R, Ganfort, Glaumax, DuoTrav, Combigan, Cosopt, Ksalakom, Ksalatan, Xalatamax, Lanotan, Latanomol, Pilotimol, Proxofelin, Prolatan, Taplotantikom, Taravatil
Terms and conditions of storage
Store at temperatures up to 25 ° C out of reach of children.
Shelf life is 2 years.
After the first opening of the bottle, it is allowed to store eye drops for no more than 4 weeks.
Terms of dispensing from pharmacies
Dispensed by prescription.
Reviews about Luxfen
Reviews about Luxfen are controversial. Many patients point to the effectiveness of the drug with increased intraocular pressure. There are also complaints about the lack of effect after a course of therapy. However, most of the messages of a negative nature are due to the development of adverse reactions, due to which the treatment had to be interrupted.
Price for Luxfen in pharmacies
The price of Luxfen for a 5 ml bottle, depending on the place of sale, can be 469-595 rubles.
Luxfen: prices in online pharmacies
|
Drug name Price Pharmacy |
|
Luxfen 0.2% eye drops 5 ml 1 pc. 309 RUB Buy |
|
Luxfen eye drops 0.2% 5ml RUB 531 Buy |

Maria Kulkes Medical journalist About the author
Education: First Moscow State Medical University named after I. M. Sechenov, specialty "General Medicine".
Information about the drug is generalized, provided for informational purposes only and does not replace the official instructions. Self-medication is hazardous to health!