- Author Rachel Wainwright wainwright@abchealthonline.com.
- Public 2023-12-15 07:39.
- Last modified 2025-11-02 20:14.
Markain
Instructions for use:
- 1. Release form and composition
- 2. Indications for use
- 3. Contraindications
- 4. Method of application and dosage
- 5. Side effects
- 6. Special instructions
- 7. Drug interactions
- 8. Analogs
- 9. Terms and conditions of storage
- 10. Terms of dispensing from pharmacies
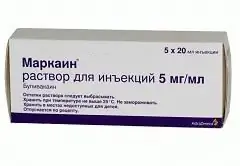
Markain is a local anesthetic drug.
Release form and composition
Markain is available in the form of a solution for injection (in 20 ml vials, 5 vials in a cardboard box).
The composition of 1 ml of injection solution contains the active substance: bupivacaine hydrochloride - 5 mg.
Indications for use
Markaine is prescribed for various types of local anesthesia (pain relief for injuries, painful diagnostic procedures (for example, arthroscopy), for surgical interventions (including cesarean section), labor pain relief):
- Conductive anesthesia, including intercostal block, block of small and large nerves, block of nerves in the neck and head;
- Regional (retrobulbar) anesthesia;
- Local infiltration anesthesia;
- Lumbar or caudal epidural block.
Contraindications
Absolute:
- Arterial hypotension;
- Diseases of the central nervous system;
- Age up to 2 years;
- Hypersensitivity to the components of the drug, as well as to local anesthetics of the amide type.
In case of accidental penetration of bupivacaine into the bloodstream, acute systemic toxic reactions may develop, therefore, the drug is not used for intravenous regional anesthesia (Beer blockade).
Relative (care must be taken for the following diseases / conditions):
- General serious condition;
- Heart block;
- Cardiovascular failure (due to the likelihood of disease progression);
- Cholinesterase deficiency;
- Inflammatory diseases or infection of the injection site (with infiltration anesthesia);
- Renal failure, decreased hepatic blood flow (eg, with liver disease, chronic heart failure);
- Septicemia, previous neurological diseases, difficulty in puncture due to spinal deformity (with caudal and lumbar anesthesia);
- Conducting paracervical anesthesia;
- Concomitant administration with antiarrhythmic drugs, including beta-blockers;
- Simultaneous use with other local anesthetics or drugs that are structurally similar to local anesthetics of the amide type (for example, lidocaine, mexiletine);
- Age under 12 and over 65;
- III trimester of pregnancy.
Method of administration and dosage
When calculating the required dose of Markain, it is important to be based on an assessment of the patient's physical status and clinical experience.
When performing prolonged blockades using prolonged infusion or fractional administration of the drug, it is necessary to take into account the possibility of increasing its concentration in blood plasma to toxic or the likelihood of developing local damage to nerve fibers. Experience has shown that administration of 400 mg of Markaine to an adult patient over 24 hours is usually well tolerated.
When using large volumes of a solution containing adrenaline, the risk of its systemic effects must be taken into account.
When choosing a dosage for adults and children from 12 years old, during the implementation of the techniques, it is most often recommended to be guided by the following data (dose / onset of action / duration of anesthesia / indications and comments):
- Infiltration: ≤30 ml, or ≤150 mg / 1-10 minutes / 3-8 hours / for surgical procedures and for postoperative analgesia;
- Retrobulbar anesthesia: 2-4 ml, or 10-20 mg / 5 minutes / 4-8 hours / used in ophthalmosurgery;
- Peribulbar anesthesia: 6-10 ml, or 30-50 mg / 10 minutes / 4-8 hours / used in ophthalmic surgery;
- Intercostal anesthesia (per nerve): 2-3 ml, or 10-15 mg / 3-5 minutes / 4-8 hours / for surgical interventions and for postoperative analgesia and pain relief in trauma;
- Interpleural block: 20 ml, or 100 mg / 10-20 minutes / 4-8 hours / for postoperative analgesia;
- Axillary supraclavicular brachial plexus block: 30-40 ml, or 150-200 mg / 15-30 minutes / 4-8 hours;
- Subclavian and interstellar perivascular brachial plexus block: 20-30 ml, or 100-150 mg / 15-30 minutes / 4-8 hours;
- Sciatic nerve block: 10-35 ml, or 50-200 mg / 15-30 minutes / 4-8 hours / during surgery;
- Blockade of the obturator nerve, femoral nerve and lateral cutaneous nerve of the thigh (3 in 1): 10-35 ml, or 50-200 mg / 15-30 minutes / 4-8 hours / during surgical interventions;
- Epidural block (lumbar level): 15-30 ml, or 75-150 mg / 15-30 minutes / 2-3 hours / during surgical interventions, including labor pain relief, cesarean section and postoperative pain relief (taking into account a trial test);
- Epidural block (chest level): 5-10 ml, or 25-50 mg / 10-15 minutes / 2-3 hours / during surgical interventions (taking into account a trial test);
- Caudal anesthesia: 20-30 ml, or 100-150 mg / 15-30 minutes / 2-3 hours / for intraoperative and postoperative anesthesia, during the treatment of pain syndromes and pain relief during diagnostic procedures (taking into account a trial test).
Markaine must be administered with caution to avoid accidental intravascular administration of the solution and the occurrence of acute toxic reactions. A thorough aspiration test is recommended before and during drug administration.
If it is necessary to administer large doses, for example, with epidural blockade, it is preliminarily recommended to conduct a test: 3-5 ml of Markain with adrenaline. In case of accidental intravascular injection of the solution, transient tachycardia may develop, easily detected by a doctor.
The main dose of Markaine should be administered slowly at a rate of 25-50 mg per minute or in fractional bolus, while maintaining constant verbal contact with the patient. If there are signs of intoxication, you must immediately interrupt the administration of the solution.
For children under 12 years of age, the dose should be calculated taking into account body weight (on average, up to 2 mg / kg):
- Lumbosacral block: 1.5-2 mg / kg;
- Thoracolumbar block: 1.5-2.5 mg / kg.
Epinephrine increases the duration of anesthesia by 50-100%.
Side effects
Adverse reactions with the use of Markain are similar to those arising from the intrathecal administration of other long-acting local anesthetics.
Side effects caused by the drug itself are difficult to distinguish from the physiological manifestations of nerve blockade (for example, bradycardia, arterial hypotension, temporary urinary retention, etc.), from reactions caused by needle insertion (indirectly (for example, epidural abscess, meningitis) or directly (for example, spinal hematoma)), as well as reactions that are associated with leakage of cerebrospinal fluid (for example, post-puncture headache).
During the use of Markaine, the following disorders can develop (> 1/10 - very often;> 1/100 and 1/1000 and <1/100 - infrequently; <1/1000 - rarely):
- Nervous system: often - paresthesia, dizziness; infrequently - symptoms of toxicity of the central nervous and cardiovascular systems (in the form of seizures, paresthesia, numbness of the tongue, visual disturbances, chills, tremors, loss of consciousness, ringing and noise in the ears, dysarthria, headache); rarely - neuropathy, unintentional total spinal block, paralysis, paraplegia, arachnoiditis;
- Cardiovascular system: very often - hypotension; often - increased blood pressure, bradycardia; rarely - cardiac arrest;
- Genitourinary system: often - urinary incontinence, urinary retention;
- Respiratory system: rarely - respiratory depression;
- Gastrointestinal tract: very often - nausea; often - vomiting;
- Organ of vision: rarely - diplopia;
- General: rarely - allergic reactions; in severe cases, anaphylactic shock.
special instructions
There is evidence of cardiac arrest or death during the administration of marcaine for peripheral blockade or epidural anesthesia. Sometimes resuscitation was difficult or impossible despite good preparation and proper anesthesia.
With high plasma concentrations of bupivacaine, cases of ventricular fibrillation, ventricular arrhythmia, sudden cardiovascular collapse and death have been reported.
Regional and local anesthesia, in addition to carrying out minor procedures, should be carried out by experienced specialists in a suitably equipped room with the availability of equipment and medicines, ready for immediate use, necessary for resuscitation and cardiac monitoring. When performing large blockades, it is recommended to insert an intravenous catheter before the introduction of Markain.
During peripheral nerve blockade, a large volume of solution is injected into an area of high vascular vascularization, often close to large vessels, which increases the risk of unintentional intravascular administration or systemic absorption of a large dose of Markain, which, in turn, can lead to an increase in plasma concentration drug.
During regional anesthesia it is necessary to be especially attentive to the following groups of patients:
- Patients receiving class III antiarrhythmic drugs (due to the possible risk of complications from the cardiovascular system, careful medical supervision is necessary);
- Weakened patients and elderly patients;
- Patients with complete or partial heart block (due to the likelihood of deterioration in myocardial conduction);
- Patients with severe renal impairment or progressive liver disease.
With some types of blockades, serious adverse reactions may develop:
- Central blockade (especially in patients with hypovolemia): suppression of the cardiovascular system is possible;
- Large peripheral blocks: large amounts of local anesthetic may be required, which may result in high plasma levels of Markain;
- Injections in the head or neck area: it is possible for the solution to enter the artery, which, even with the introduction of low doses, can lead to serious adverse reactions;
- Retrobulbar and peribulbar administration: there is a risk of persistent dysfunction of the eye muscles. The severity of these reactions is determined by the degree of injury, the concentration of Markaine, and the duration of tissue exposure to the drug. It is recommended to use the minimum effective concentration and dose of the solution. With simultaneous use with vasoconstrictors and other additives, it is possible to increase tissue reactions, therefore, they must be used strictly according to indications;
- Paracervical blockade: the development of tachycardia and / or bradycardia in the fetus is possible, and therefore it is necessary to carefully monitor the fetal heart rate;
- Retrobulbar injections: Markaine can accidentally get into the cranial subarachnoid space, which can lead to the development of temporary blindness, apnea, seizures, collapse and other side effects (the resulting disorders should be diagnosed and stopped in a timely manner);
- Epidural anesthesia: Hypotension and bradycardia may develop. To reduce the likelihood of developing these complications, crystalloid and colloidal solutions are preliminarily administered. If hypotension occurs, intravenous administration of sympathomimetics is indicated (if necessary, repeat administration). In children, the doses used should be appropriate for weight and age.
The Markain solution does not contain preservatives, so it should be used immediately after opening the bottle.
Drug interactions
With the simultaneous use of Markain with certain drugs, the following effects may occur:
- Other local anesthetics or drugs that have a structure similar to local anesthetics of the amide type (for example, lidocaine, mexiletine): development of an additive toxic effect (care should be taken when using a combination of these drugs);
- Tricyclic antidepressants, monoamine oxidase inhibitors: increased risk of severe high blood pressure;
- Preparations containing oxytocin or ergotamine: the development of a steady increase in blood pressure with the development of further complications from the cardiovascular system;
- Inhalation anesthesia with halothane: increased risk of arrhythmias;
- Disinfectant solutions containing heavy metals (when processing the injection site): an increase in the risk of developing local reactions in the form of edema and pain;
- Drugs that have a similar structure to local anesthetics (for example, tocainide): increased risk of developing an additive toxic effect;
- Drugs that depress the central nervous system: increasing its suppression.
The solubility of Markain decreases at pH> 6.5, which must be taken into account when adding alkaline solutions (due to the possible formation of precipitate).
Analogs
Markain's analogues are: Markain Spinal, Markain Spinal Heavy, Buvanestin, Bupivacaine, Bupikain, BlokkoS.
Terms and conditions of storage
Keep out of reach of children at temperatures up to 25 ° C, do not freeze.
The shelf life is 3 years.
Terms of dispensing from pharmacies
Dispensed by prescription.
Information about the drug is generalized, provided for informational purposes only and does not replace the official instructions. Self-medication is hazardous to health!