- Author Rachel Wainwright wainwright@abchealthonline.com.
- Public 2023-12-15 07:39.
- Last modified 2025-11-02 20:14.
Mitomycin-S Kiova
Instructions for use:
- 1. Release form and composition
- 2. Indications for use
- 3. Contraindications
- 4. Method of application and dosage
- 5. Side effects
- 6. Special instructions
- 7. Drug interactions
- 8. Analogs
- 9. Terms and conditions of storage
- 10. Terms of dispensing from pharmacies
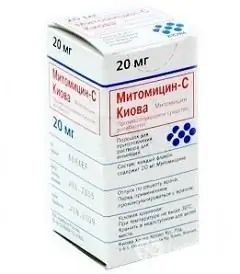
Mitomycin-S Kiova is an antitumor antibiotic.
Release form and composition
Dosage form - crystalline powder (or crystals) for the preparation of a solution for injection, blue-violet (2, 10 and 20 mg in vials made of colorless transparent glass; in a cardboard box with cells 10 vials (2 mg); in a cardboard box 5 vials packed in individual cardboard boxes (10 and 20 mg each)).
Active ingredient: mitomycin, in 1 bottle - 2, 10 or 20 mg.
The auxiliary component is sodium chloride.
Indications for use
- Esophageal carcinoma;
- Stomach cancer;
- Colon and rectal cancer;
- Bile duct cancer;
- Liver cancer;
- Pancreas cancer;
- Cervical cancer;
- Vulvar cancer;
- Endometrial cancer;
- Mammary cancer;
- Mesothelioma;
- Bladder cancer;
- Cancer of the renal pelvis and ureters;
- Prostate cancer;
- Non-small cell lung cancer;
- Malignant tumors of the neck and head.
Contraindications
Absolute:
- Severe renal dysfunction;
- Severe depression of bone marrow function;
- Increased bleeding or bleeding disorders;
- Pregnancy;
- Lactation;
- Hypersensitivity to mitomycin.
Relative (special care should be taken due to the risk of complications):
- Childhood;
- Acute infectious diseases of bacterial, fungal or viral origin.
Method of administration and dosage
Mitomycin-S Kiova is injected slowly intravenously or intravesically (in the treatment of bladder tumors), if necessary - intraperitoneally, intrapleurally or intraarterially. Before administration, the powder is dissolved with water for injection to a concentration of 0.4 mg / ml.
Doses are set by the doctor individually, depending on the indications, the general condition of the patient and the optimal scheme of anticancer therapy.
Usually the drug is prescribed in the following doses:
- 20 mg / m 2 intravenously 1 time every 4-6 weeks;
- 2 mg / m 2 intravenously 1 time in 1-5 and 8-12 days every 4-6 weeks;
- 10 mg / m 2 intravenously (in combination with other cytostatics) 1 time every 6-8 weeks;
- 30-40 mg, diluted with 30-60 ml of sterile aqueous solution (concentration not more than 1 mg / ml), intravesical once a week for 6-8 weeks, then once a month for 6 months.
Due to the likelihood of cumulative myelosuppression caused by mitomycin, with subsequent intravenous injections, the dose of the drug is adjusted depending on the state of bone marrow function.
Recommended doses depending on the minimum blood count after the first dose (in 1 ml):
- Leukocytes over 3000 and / or platelets over 75000 - a dose similar to the first;
- Leukocytes 2000-2999 and / or platelets more than 25 000-74 999 - 70% of the previous dose;
- Leukocytes less than 2000 and / or platelets less than 25,000 - 50% of the previous dose.
If, before starting the next course of therapy, blood counts have recovered (ie leukocytes have reached 4000 / mm 3 and platelets - 100,000 / mm 3 blood), Mitomycin-S Kiova can be administered in doses adjusted in accordance with the table.
If the drug is prescribed in combination with other myelosuppressive drugs, the dose of mitomycin should be adjusted accordingly.
Side effects
- Hematopoietic system: thrombocytopenia, leukopenia, anemia;
- Respiratory system: dry cough, shortness of breath, lung infiltrates;
- Urinary system: an increase in the concentration of creatinine in the blood, the development of hemolytic uremic syndrome, accompanied by microangiopathic hemolytic anemia with fragmentation of erythrocytes and anuric form of acute renal failure, as well as thrombocytopenia; rarely - pulmonary edema, increased blood pressure, neuropathy, syncope, hemolytic anemia (in patients who received mitomycin intravenously as a monopreparation or in combination with other cytostatics at doses over 60 mg);
- Digestive system: stomatitis, liver dysfunction, esophagitis, diarrhea, anorexia, nausea, vomiting;
- Cardiovascular system: development or worsening of heart failure (in patients previously treated with doxorubicin), decrease in myocardial contractility;
- Skin and its appendages: purple streaks on the nails, skin rash or ulceration, reversible alopecia;
- Others: increased weakness or fatigue, tingling or numbness in the fingers and toes, increased body temperature;
- Local reactions: thrombophlebitis, rash and itching in the genitals and hands; if the solution gets under the skin - pain, redness, inflammation of the subcutaneous fat and necrosis; with intravesical administration - increased frequency of urination, nocturnal enuresis, cystitis, dysuric disorders, urinary tract irritation, bladder atrophy, hematuria and other symptoms of local irritation.
special instructions
Treatment with Mitomycin-S Kiova should be carried out under the supervision of a medical specialist with experience in working with anticancer drugs.
Intravenous administration should be carried out slowly, with extreme caution to prevent the drug from entering the extravasal space.
The entire course of treatment and within 8 weeks after its completion, it is necessary to monitor the parameters of peripheral blood (the amount of hemoglobin, platelets, leukocytes and neutrophils), as well as the concentration of urea and creatinine in the blood serum.
For 3 months after the end of the course of anticancer therapy, all patients (both women and men) should use reliable methods of contraception.
Drug interactions
Patients previously treated with doxorubicin may develop congestive heart failure during treatment with mitomycin.
Increasing the toxicity of mitomycin is possible with simultaneous radiation therapy or the use of drugs that have nephrotoxic and myelotoxic effects.
In the case of preliminary or simultaneous administration of vinca alkaloids with mitomycin, the development of respiratory distress syndrome is possible. This syndrome was also observed in patients who received oxygen therapy (inhalation of a mixture containing more than 50% oxygen) during the period of mitomycin use.
Analogs
Analogues of the drug Mitomycin-S Kiova are: Vero-Mitomycin, Mitomycin C, Mutamycin, Mitomycin-Lance.
Terms and conditions of storage
Store at temperatures up to 30 ° C out of reach of children.
Shelf life is 4 years.
Terms of dispensing from pharmacies
Dispensed by prescription.
Information about the drug is generalized, provided for informational purposes only and does not replace the official instructions. Self-medication is hazardous to health!