- Author Rachel Wainwright wainwright@abchealthonline.com.
- Public 2023-12-15 07:39.
- Last modified 2025-11-02 20:14.
Avaxim 80
Instructions for use:
- 1. Release form and composition
- 2. Indications for use
- 3. Contraindications
- 4. Method of application and dosage
- 5. Side effects
- 6. Special instructions
- 7. Drug interactions
- 8. Analogs
- 9. Terms and conditions of storage
- 10. Terms of dispensing from pharmacies
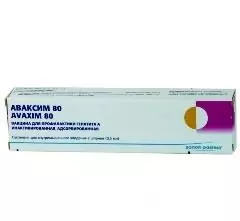
Avaxim 80 is a vaccine for the prevention of viral hepatitis A in children.
Release form and composition
Avaxim 80 is produced in the form of a suspension for intramuscular administration: a turbid whitish liquid (1 dose in glass syringes of type 1 with a capacity of 1 ml with a fixed stainless steel needle with a protective cap and an elastomer piston, 1 syringe in a transparent closed cell package (blister), 1 package (blister) in a cardboard box).
The composition of 1 dose (0.5 ml) of the suspension includes:
- Active substance: inactivated hepatitis A virus - 80 U (units, expressed using the manufacturer's internal reference standard);
- Auxiliary components: aluminum hydroxide - 0.15 mg (in terms of aluminum), formaldehyde - 0.0125 mg, 2-phenoxyethanol - 0.0025 ml, Hanke 199 medium (a mixture of amino acids, mineral salts, vitamins and other components dissolved in water for injection) - up to 0.5 ml, hydrochloric acid or sodium hydroxide - in the amount necessary to adjust the pH value.
Indications for use
Avaxime 80 is used for the specific prevention of viral hepatitis A in children aged 1-15 years (inclusive).
Contraindications
Absolute:
- Diseases proceeding with fever, acute or chronic infectious diseases in the exacerbation stage (vaccination is possible 2-4 weeks after recovery or during remission or convalescence; with a mild course of acute respiratory viral infections, with acute intestinal infections, vaccinations are carried out immediately after the temperature has returned to normal);
- Systemic hypersensitivity reactions or life-threatening reactions to the previous administration of Avaxim 80 or a vaccine containing similar components;
- Allergy to drug components.
Relative (the use of Avaxim 80 is possible only with caution in the presence of the following diseases / conditions):
- Thrombocytopenia or disorders of the blood coagulation system (due to the likelihood of bleeding after intramuscular injection). After injection, such patients need to apply a pressure bandage for at least 2 minutes. In exceptional cases, subcutaneous administration of the drug is possible;
- Hypersensitivity to formaldehyde, neomycin or other antibiotic drugs of this class;
- Pregnancy and lactation period (the use of Avaxim 80 in pregnant women is not recommended (due to the lack of the necessary data); the decision on vaccination should be made only if there are unambiguous indications after the benefit-risk ratio).
Before vaccination, a healthcare professional must take all measures necessary to prevent the development of anaphylactoid or anaphylactic reactions at the time of vaccination (the necessary medicines, including epinephrine, must be available).
A weak immune response to the vaccine can be caused by immunodeficiency states or immunosuppressive therapy. In these cases, it is recommended to postpone vaccination until recovery or treatment ends. Nevertheless, Avaxim 80 is prescribed for patients with chronic immunodeficiencies, such as HIV infection, even in cases where the immune response to its administration may be reduced due to the underlying disease.
The introduction of a needle can provoke psychogenic fainting (in the form of weakness, loss of consciousness); therefore, vaccinations should be carried out in conditions that prevent injury from a fall and provide the necessary medical attention.
Method of administration and dosage
The drug is injected intramuscularly into the deltoid muscle of the shoulder, for children under 2 years of age - into the upper outer region of the thigh. The suspension should not be injected into the gluteal region or intradermally (possibly weakening of the immune response). Introduction into the vascular bed is strictly prohibited (make sure that the needle does not enter the blood vessel).
The syringe with Avaxim 80 should be shaken well before vaccination until a homogeneous suspension is obtained.
Single dose - 0.5 ml.
Mixing Avaxim 80 in one syringe with other vaccines is prohibited!
In order to ensure long-term immunity, revaccination should be carried out with the same dose after 6 months or up to 3 years after the introduction of the first dose.
In immunocompetent individuals, there is no need for revaccination after double vaccination.
The vaccine should not be used in cases of discoloration or in the presence of foreign particles. Any unused vaccine residue is discarded.
Side effects
All disorders identified as a result of clinical trials were of a mild, short-term nature and resolved on their own without additional treatment.
Possible adverse reactions (> 10% - very common;> 1% and 0.1% and 0.01% and <0.1% - rarely; <0.01% - very rare; with an unknown frequency - if it is impossible to estimate the frequency development of violations according to available data):
- Nervous system: often - headache;
- Musculoskeletal and connective tissue: often - arthralgia, myalgia;
- Gastrointestinal tract: often - diarrhea, abdominal pain, nausea, vomiting;
- Skin and subcutaneous tissue: infrequently - rash, urticaria;
- Psyche: often - irritability, insomnia;
- Metabolism and nutrition: often - decreased appetite;
- Violations at the injection site and general disorders: often - mild soreness, redness, induration and swelling at the injection site, weakness, moderate fever.
Violations were reported less frequently after revaccination than after primary vaccination. In hepatitis A seropositive patients, vaccination was as well tolerated as in seronegative patients.
The reactions seen in children with hemophilia are similar to those seen in adults.
As a result of post-marketing observations, it was also found that as a result of the use of Avaxim 80, vasovagal syncope may develop (with an unknown frequency).
With the development of any side effects (both described and not described in the instructions), you should consult your doctor.
special instructions
Immunization is especially recommended for the following groups of people:
- Living in regions, unfavorable for the incidence of hepatitis A;
- Traveling to disadvantaged regions (countries), where an outbreak of hepatitis A is recorded;
- Those in contact with patients in the foci of hepatitis A.
Also, vaccination can be given to children who do not have an increased risk of infection, in cases where their legal representatives (guardians) want to protect them from hepatitis A.
Avaxim 80 does not provide protection against hepatitis caused by other pathogens (hepatitis B, C, E), as well as other known pathogens that affect the liver.
Seropositivity for hepatitis A is not a contraindication to vaccination.
Immunization may not be effective in infected patients vaccinated during the incubation period for hepatitis A.
The doctor must be warned about the recent or coinciding with the vaccination use of any other drug, including non-prescription drugs.
Drug interactions
Avaxime 80 can be administered simultaneously to different parts of the body with vaccines that are included in the National Calendar of Preventive Vaccinations and contain the following components (one or more): toxoids - diphtheria, tetanus; vaccines - against viral hepatitis B, pertussis (whole cell or acellular), measles, hemophilic type b, polio (inactivated or live), against endemic mumps and rubella.
Since the vaccine is inactivated, its combined use with other inactivated vaccines, as a rule, subject to the introduction of vaccines in different parts of the body, does not affect the effectiveness of vaccination.
The drug can be administered simultaneously with hepatitis A immunoglobulin (in different parts of the body). This does not affect the rate of seroconversion, but may lead to the production of lower antibody titers.
Avaxime 80 can be used as a booster dose in cases where another inactivated hepatitis A vaccine was used for the primary vaccination.
Analogs
The analogues of Avaxim 80 are: Avaxim 160, Vakta, Havrix.
Terms and conditions of storage
Store in a dark place, out of reach of children, at a temperature of 2-8 ° C, do not freeze.
The shelf life is 3 years.
Terms of dispensing from pharmacies
Dispensed by prescription
Information about the drug is generalized, provided for informational purposes only and does not replace the official instructions. Self-medication is hazardous to health!